Constellation Technologies’ Cold Chain solution has been deployed to monitor the temperature integrity of Pfizer vaccine doses at Barwon Health’s University Hospital Geelong, one of the initial sites in Australia storing vaccines. CT’s proprietary Cold Chain solution is now deployed across all Barwon Health departments and facilities in more than 200 refrigeration assets.
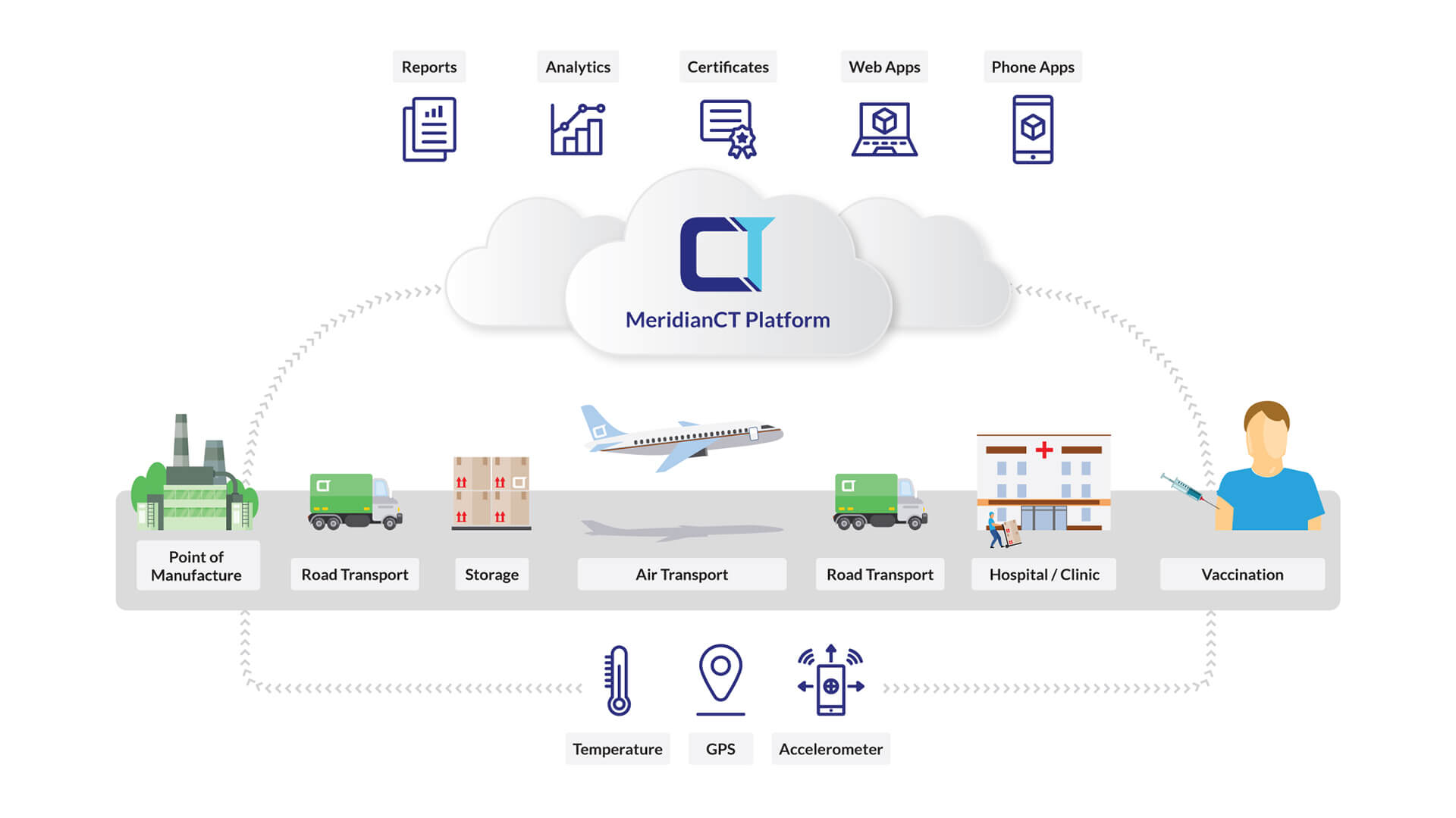
The ‘Cold Chain’ is a system of transporting and storing temperature sensitive products such as vaccines. The Cold Chain begins from the time the vaccine is manufactured, continues through to the vaccine distribution centre across trucking, flights, warehousing, pharmacies as well as the interchanges at each point, and only ends when the vaccine is administered.
The CT Cold Chain Solution comprises of sensors that travel with the vaccine doses on their journey through-out the cold chain and continuously tracks temperature, location and handling. Multiple sensors communicate with the one central software platform – CT’s core IoT platform – The MeridianCT (MCT) Platform.
Complete and continuous monitoring is required to verify that the integrity, and thus the safety and efficacy, of the vaccine has not been compromised in transit and storage due to temperature breaches (too hot or too cold) and handling.
According to a previous World Health Organization paper, around half of the vaccines distributed in the world go to waste, in large part because of a failure of the cold chain.1
The challenges that any potential Cold Chain monitoring solution must address to adequately monitor cold chain integrity are listed and addressed through the following features of the CT Cold Chain solution:
- Recognised and verified accuracy at extreme temperatures
- Handling: including shock and location tracking
- Capacity of the software platform to receive and process millions of data records
- Real-time customised alerts and reporting
- Storing and transmitting the data especially during sensitive transit windows such as custodian changes to maintain a complete historical and automated and independent verification record
- Connectivity across multiple communications networks to ensure communication stability
- Predictive analytics to identify potential cold chain failures and thus allow proactive action to be taken to avoid cold chain breaches
- Dynamic reporting and viewing platforms to communicate with multiple stakeholders
- Compliance with data security standards
- Scalability of hardware production and platform operational capacity to support global deployment.
- Independent power supply and operation of data capture and transmission
Through the global COVID-19 vaccination program under which 9 billion doses are expected to be produced in 2021 alone, the importance of cold chain integrity has become a universal concern with growing community awareness. The MCT platform can provide the third-party data and reporting to verify the success or failure at every point in the Cold Chain.
The solution has been operational for five years and deployed in multiple countries. It is one of the very few IoT solutions that has undergone NATA (National Accreditation and Testing Authority) calibration to confirm its ability to monitor extreme temperatures to the requisite accuracy for highly sensitive materials such as vaccines.
In the health sector, the CT Cold Chain Solution currently services a number of hospitals and research facilities in Australia and is working closely with cold chain stakeholders on imminent further deployments to support the rollout of the COVID-19 vaccines.
1 https://apps.who.int/iris/bitstream/handle/10665/68463/WHO_VB_03.18.Rev.1_eng.pdf
CT Monitoring Solution – Summary of key features
Hardware Features
- Standalone System
- No Power Supply (Long lasting rechargeable battery –5500mah ~1yr)
- IP66
- CAT-M1 (Telstra/Vodafone) / LoraWan / Sigfox
- In Built Sensor (-30C to 50C)
- Plug in Probe (-80C to 200C)
- Plug in Humidity Sensor
Web/Mobile Apps
- Push Notification
- Continuous near real time dashboards (traffic Light based alerting)
- Centralised group monitoring supporting ‘3 levels’ organisational hierarchy
- Alert escalations
Artificial Intelligence
- Models for predicting failures, anomaly detection.
- Door status alerts
- Compressor failure
Transportation
- GPS Location based dashboards.
- Accelerometers (Shock Detection)
- Geofence reports
- Route monitoring
Digital forms
- Complete digital record keeping using mobile apps.
- Convert all paper-based forms to electronic form.
- Monitor submissions and create action list based on form submission.
- Auto emailing of reports – Compliance, daily reporting, verification certificates
- Staff task assignment.
- Staff KPI’s
- Asset Management – Equipment maintenance records
Compliance
- HACCP compliance temperature reports
- Corrective actions
- Alert actions
- Customised compliance reports